 
Remember, the solid and liquid phases of H 2O can coexist at 0☌. Solid has the least energetic state atoms in solids are. Changes like these occur when the system is given or taken a large amount of energy, or when the pressure on it is altered. The three phases of matter that you frequently encounter are solid, liquid and gas (see Figure 11.9). If heat is added to ice at 0☌, some of the solid changes phase to make liquid, which is also at 0☌. States of Matter: Basics - Phase Changes Estimated Duration: 10 minutes Heat, cool, and compress atoms and molecules and watch as they change between solid. Changes in a matters state (from solid to liquid to gas or plasma) are known as phase changes in scientific terms. These changes occur when sufficient energy is supplied to the system (or a sufficient amount is lost), and also occur when the pressure on the system is changed. When liquid water ( H 2 O) freezes into a solid state (ice), it appears changed however, this change is only physical, as the composition of the constituent molecules is the same: 11.19 hydrogen and 88.81 oxygen by mass. In other words, phase changes are isothermal (isothermal means “constant temperature”). A phase change is when matter changes to from one state (solid, liquid, gas, plasma) to another. As long as you are at 100 C, you can change the phase by changing the pressure on the system. 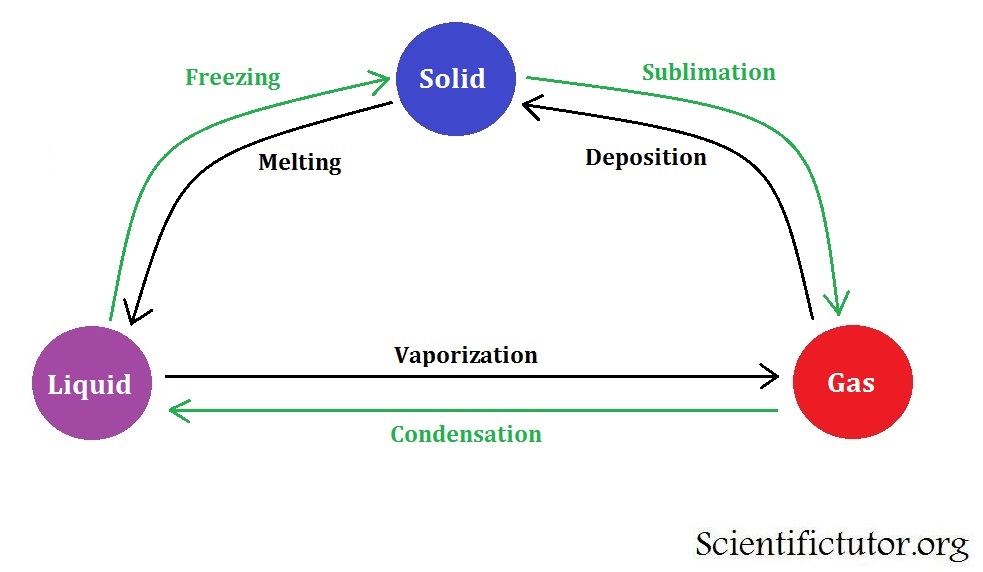
A phase change is the process that can alter the forms of matter. Imagine a vertical line through this diagram- for water, choose 100 degrees C. Matter has forms, such as a liquid, solid, or a gas. /phase-changes-56a12ddd3df78cf772682e07.png)
Its sublimation temperature at atmospheric pressure is \(-78^oC\).\), the temperature of a substance does not change as the substance goes from one phase to another. In the video here, Sal uses a horizontal line through the phase diagram. Solid \(CO_2\) is known as dry ice because instead of melting, it sublimes. Examples of phase changes are melting (changingfrom a solid to a. Carbon dioxide has no liquid phase at atmospheric pressure. Whentemperature changes, matter can undergo a phase change, shifting fromone form to another. 
\) shows the result, as well as showing a familiar example of sublimation. A phase change is the reversible physical change that occurs when a substance changes from one state of matter to another. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
